The Evolution of Birds
The Evolution of Birds: From Theropod Dinosaurs to Masters of the Air
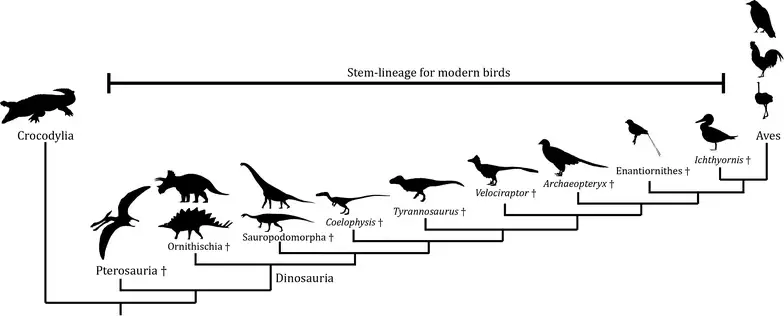
The evolution of birds represents one of the most compelling and well-documented transitions in the history of life: the transformation of small, ground-dwelling theropod dinosaurs into the highly specialized, volant organisms we recognize today. This evolutionary story is not a simple linear progression but rather a branching, mosaic process involving anatomical innovation, ecological opportunity, and deep-time experimentation with form and function.
Theropod Origins: Birds as Living Dinosaurs
Modern birds (class Aves) are now universally accepted as the descendants of theropod dinosaurs, specifically within the clade Maniraptora. This conclusion, once controversial, rests on a convergence of evidence from comparative anatomy, cladistics, and, more recently, molecular data. Birds share a remarkable suite of characteristics with theropods: hollow (pneumatized) bones, three-toed limbs, a furcula (wishbone), posteriorly directed pubis, and highly modified wrists that allow folding of the forelimb.
Particularly significant are dromaeosaurids and troodontids—groups of small, often feathered theropods that exhibit numerous bird-like traits. Fossils from the Late Jurassic and Early Cretaceous, especially those discovered in the Liaoning deposits of northeastern China, have revealed an extraordinary diversity of feathered dinosaurs. These specimens demonstrate that feathers did not originate with birds per se, but rather evolved earlier within theropods.
The Origin of Feathers
Feathers are among the most defining characteristics of birds, yet their evolutionary origin predates powered flight. Early feather-like structures appear to have been simple, filamentous integumentary appendages—perhaps initially serving thermoregulatory or display functions. Over time, these structures became increasingly complex, branching into barbs and barbules that form the interlocking vanes of modern feathers.
Developmental biology supports this sequence, showing that feather morphogenesis can proceed through stages that mirror these evolutionary steps. Importantly, many non-avian theropods possessed feathers or feather-like coverings, including forms such as Sinosauropteryx, which bore simple filaments, and Microraptor, which exhibited fully developed pennaceous feathers on both forelimbs and hindlimbs.
The presence of aerodynamic feathers in non-avian dinosaurs suggests that flight-related structures evolved before true flight, a key example of exaptation—where a trait evolves for one function and is later co-opted for another.
Archaeopteryx and the Mosaic Transition
The Late Jurassic genus Archaeopteryx remains one of the most iconic transitional fossils in evolutionary biology. Possessing both avian and reptilian features, Archaeopteryx had well-developed flight feathers and wings, yet retained teeth, a long bony tail, and clawed fingers.
Rather than representing a direct ancestor of modern birds, Archaeopteryx is better viewed as part of a broader radiation of early avialans—bird-like dinosaurs experimenting with various locomotor and ecological strategies. Its anatomy suggests that it was capable of some form of flight, though whether this was powered flapping flight or primarily gliding remains debated.
The Evolution of Flight
The origin of flight in birds has been the subject of extensive debate, traditionally framed as a dichotomy between the “trees-down” (arboreal) and “ground-up” (cursorial) hypotheses. Increasingly, evidence suggests that this binary framing may be overly simplistic.
Forms like Microraptor, with its four-winged morphology, support the idea that early flight may have involved gliding or parachuting from elevated positions. Conversely, certain anatomical features in theropods—such as strong hindlimbs and feathered forelimbs—are consistent with running-based models where proto-wings aided in balance, maneuverability, or insect capture.
A synthesis view has gained traction: flight likely evolved through a series of intermediate behaviors, including wing-assisted incline running, controlled descent, and eventually powered flapping. The incremental enlargement of the forelimbs, reorientation of the shoulder joint, and development of asymmetrical flight feathers all contributed to the emergence of true flight.
Skeletal and Physiological Innovations
The transition to flight required extensive modifications to the avian body plan. The skeleton became highly specialized for both strength and lightness. Bones became pneumatized, reducing weight while maintaining rigidity. The fusion of elements—such as the formation of the synsacrum, pygostyle, and carpometacarpus—enhanced structural stability.
The sternum developed a keel (carina) to anchor powerful flight muscles, particularly the pectoralis and supracoracoideus. The latter, through a pulley-like arrangement involving the triosseal canal, allows the upstroke of the wing—an innovation critical for sustained flapping flight.
Respiratory efficiency also underwent dramatic enhancement. Birds possess a unique system of air sacs that enables unidirectional airflow through the lungs, ensuring continuous gas exchange during both inhalation and exhalation. This system likely evolved in theropod ancestors and may have initially been associated with high metabolic demands rather than flight per se.
The Beak and Loss of Teeth
Another major transformation in avian evolution is the loss of teeth and the development of a keratinous beak. Early birds, including Archaeopteryx and Ichthyornis, retained teeth, but these were gradually lost in the lineage leading to modern birds (Neornithes).
The beak is an extraordinarily versatile structure, capable of evolving rapidly in response to ecological pressures. Its diversity in shape and function—from the probing bill of shorebirds to the crushing beak of finches—has played a central role in avian adaptive radiation.
The loss of teeth may have reduced weight and facilitated faster developmental rates, though the exact selective pressures remain a topic of ongoing research.
The Cretaceous Radiation and Mass Extinction
By the Late Cretaceous, birds had diversified into several major lineages, including Enantiornithes and Ornithuromorpha. The former, now extinct, were highly successful and ecologically diverse, while the latter includes the ancestors of modern birds.
The end-Cretaceous mass extinction event, approximately 66 million years ago, marked a pivotal moment. While non-avian dinosaurs were wiped out, a subset of avian lineages survived. The reasons for this differential survival are still debated but may include factors such as small body size, ecological flexibility, and possibly seed-based diets that buffered against post-impact ecological collapse.
Following the extinction, birds underwent a rapid adaptive radiation during the Paleogene, filling ecological niches left vacant by the disappearance of other vertebrates.
Modern Diversification
Today, birds comprise over 11,000 species, occupying nearly every terrestrial and many aquatic environments on Earth. Their evolutionary success is attributable not only to flight but also to their behavioral flexibility, advanced sensory systems, and complex social behaviors.
Genomic studies have further refined our understanding of avian evolution, revealing patterns of rapid diversification and convergent evolution across lineages. The integration of fossil and molecular data continues to reshape the avian phylogenetic tree, resolving long-standing questions while raising new ones.
Conclusion
Bird evolution is not merely a story of flight but of transformation—of how a lineage of small theropod dinosaurs acquired feathers, refined their physiology, and ultimately took to the skies. It is a testament to the power of natural selection and the intricate interplay between form, function, and environment.
Far from being separate from dinosaurs, birds are their living descendants—dynamic, diverse, and still evolving. In every sparrow and eagle, we see not just a bird, but the enduring legacy of a deep and remarkable evolutionary history.